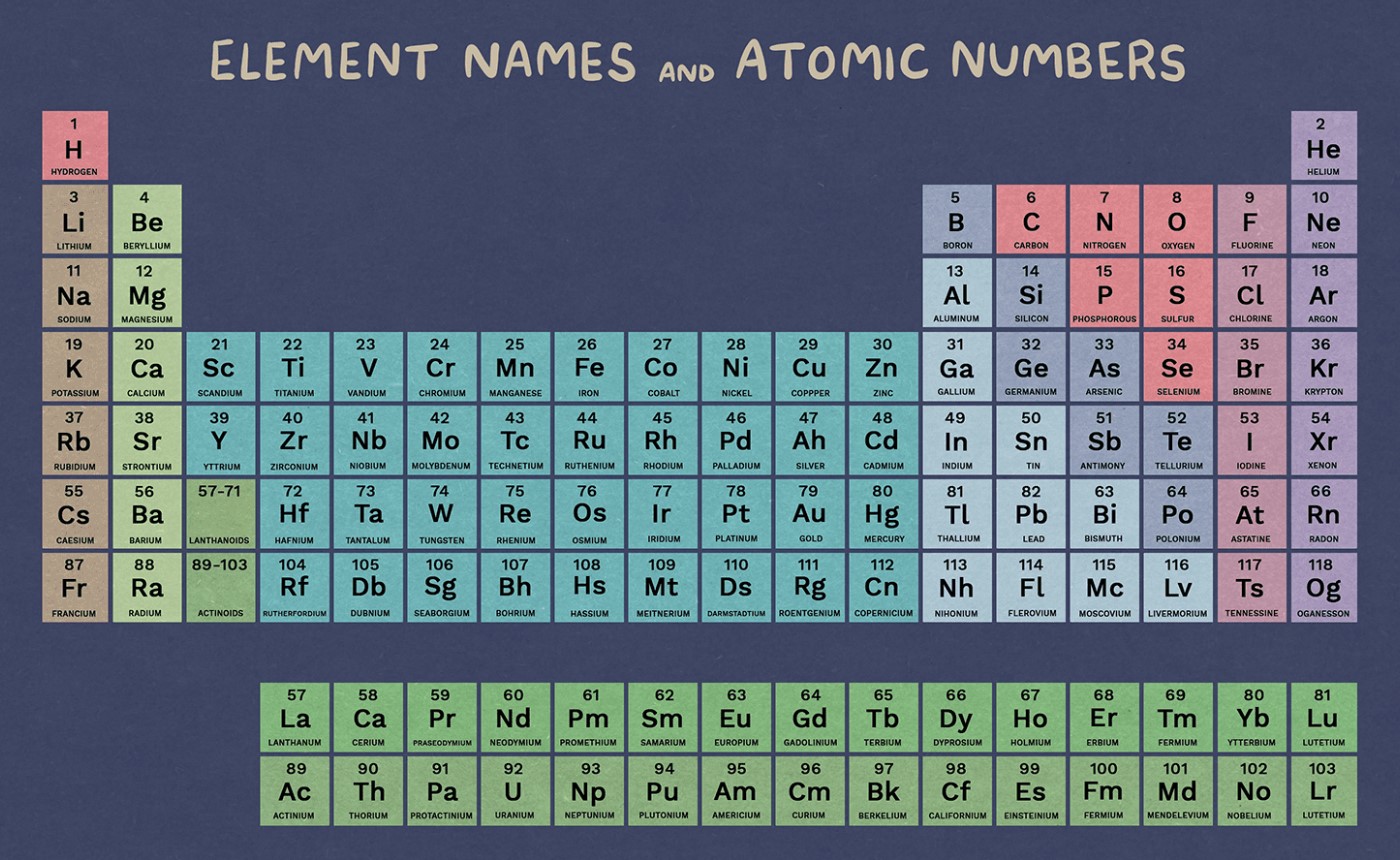
What is a Periodic Table?
The chemical elements are arranged in rows and columns on what is known as the periodic table, or the periodic table of the (chemical) elements. It is frequently used in physics, chemistry, and other sciences and is frequently regarded as a symbol of chemistry. It is a visual representation of the periodic law, which claims that the atomic numbers of chemical elements have a roughly periodic relationship with their attributes. The table is divided into four blocks that are rectangular in shape. The table’s columns are referred to as groups and its rows are known as periods. The periodic table’s elements in the same group have comparable chemical properties.
Trends can be seen throughout the periodic table, with metallic characteristics (surrendering electrons to other atoms) increasing in the opposite way from left to right across a period and from down to up across a group. The configurations of an atom’s electrons are the primary cause of these patterns.
Atoms with an equal number of positively charged protons and negatively charged electrons are only listed in the periodic table along with isotopes, which are atoms with the same number of protons but differing quantities of neutrons. Various tables, such as the tables of nuclides, graphically compile other atoms, such as nuclides and isotopes.
Why Was the Periodic Table Created?
The periodic table that we use today is an improved version of a few models that were put forth by scientists in the 19th and 20th centuries. Dimitri Mendeleev proposed the periodic table based on the findings of the scientists who came before him, such as John Newlands and Antoine-Laurent de Lavoisier. The periodic table was created by Mendeleev, who is the only person to receive credit for it.
Why Are Elements Arranged in a Table?
Although it is as commonplace to see the chemical elements arrayed in the periodic table today as it is to see a map of the globe, this was not always the case.
Dmitri Mendeleev, the inventor of the periodic table, started gathering and organising known properties of elements in 1869 while he was travelling by train. While he observed that certain sets of elements displayed similar characteristics, he also noted that there were many exceptions to the patterns that were beginning to emerge.
Amazingly, rather than abandoning up, he attempted to change the measured property values to better match the patterns! To help the patterns in his “game” work out, he also anticipated that certain elements must exist that weren’t there at the time.
Although it took years to win over the world’s critics, Mendeleev’s patterns could not be disproved once freshly found elements matched those he had anticipated. Additionally, after recalculating several of the attributes he “fudged,” it was discovered that they were substantially closer to his expectations.
Why did the Mendeleev Periodic Table get so much traction?
The earliest version of the periodic table, which is close to the one we currently use, was proposed by Dimitri Mendeleev, who is sometimes referred to as the father of the periodic table. The primary distinction between Mendeleev’s periodic law and the current periodic law is this.
While the present periodic law is based on the ascending order of atomic numbers, Mendeleev modelled his periodic table on the basis of increasing atomic mass.
Mendeleev was able to foresee the discovery and characteristics of several elements despite the fact that his periodic table was based on atomic weight. Only roughly half of the elements that we now know existed during his time, and the majority of what was known about them was erroneous. The German Journal of Chemistry first published Mendeleev’s Periodic Table in 1869.
First 20 Elements of the periodic table in order
- H – Hydrogen
- He – Helium
- Li – Lithium
- Be – Beryllium
- B – Boron
- C – Carbon
- N – Nitrogen
- O – Oxygen
- F – Fluorine
- Ne – Neon
- Na – Sodium
- Mg – Magnesium
- Al – Aluminium
- Si – Silicon
- P – Phosphorus
- S – Sulphur
- Cl – Chlorine
- Ar – Argon
- K – Potassium
- Ca – Calcium
What does the contemporary periodic table describe?
The current periodic law serves as the foundation for the periodic table’s lengthy form. The elements are organised in the table in ascending order of their atomic numbers. The term “modern periodic table” refers to the periodic table as it now stands. The building is composed of 18 vertical columns and 7 horizontal rows..
Key Terms from the Periodic Table
- Atomic Number: An element’s atomic number is the total number of protons in its nucleus (Z). For instance, since carbon has six protons, its atomic number must only be six.
- Atomic mass is defined as the typical mass of an element’s atoms. It is calculated using the relative natural abundance of the element’s isotopes. Atomic weight is another name for atomic mass (A). It is expressed in atomic masses (AMU).
- Periods are the horizontal rows in a periodic table that go from left to right. The periodic table has seven different periods in all. Due to the similarities in their physical characteristics, such as having the same atomic orbitals, the elements are stacked horizontally.
- Group: In a periodic table, the vertical columns from top to bottom are referred to as Groups. The periodic table contains 18 groups in all. Because the elements have comparable characteristics and have the same amount of valence electrons, they are placed vertically.
- Elements’ symbols: Using a letter or a group of two to three letters, a chemical element can be represented by a symbol. e.g. Symbol for a chemical.



